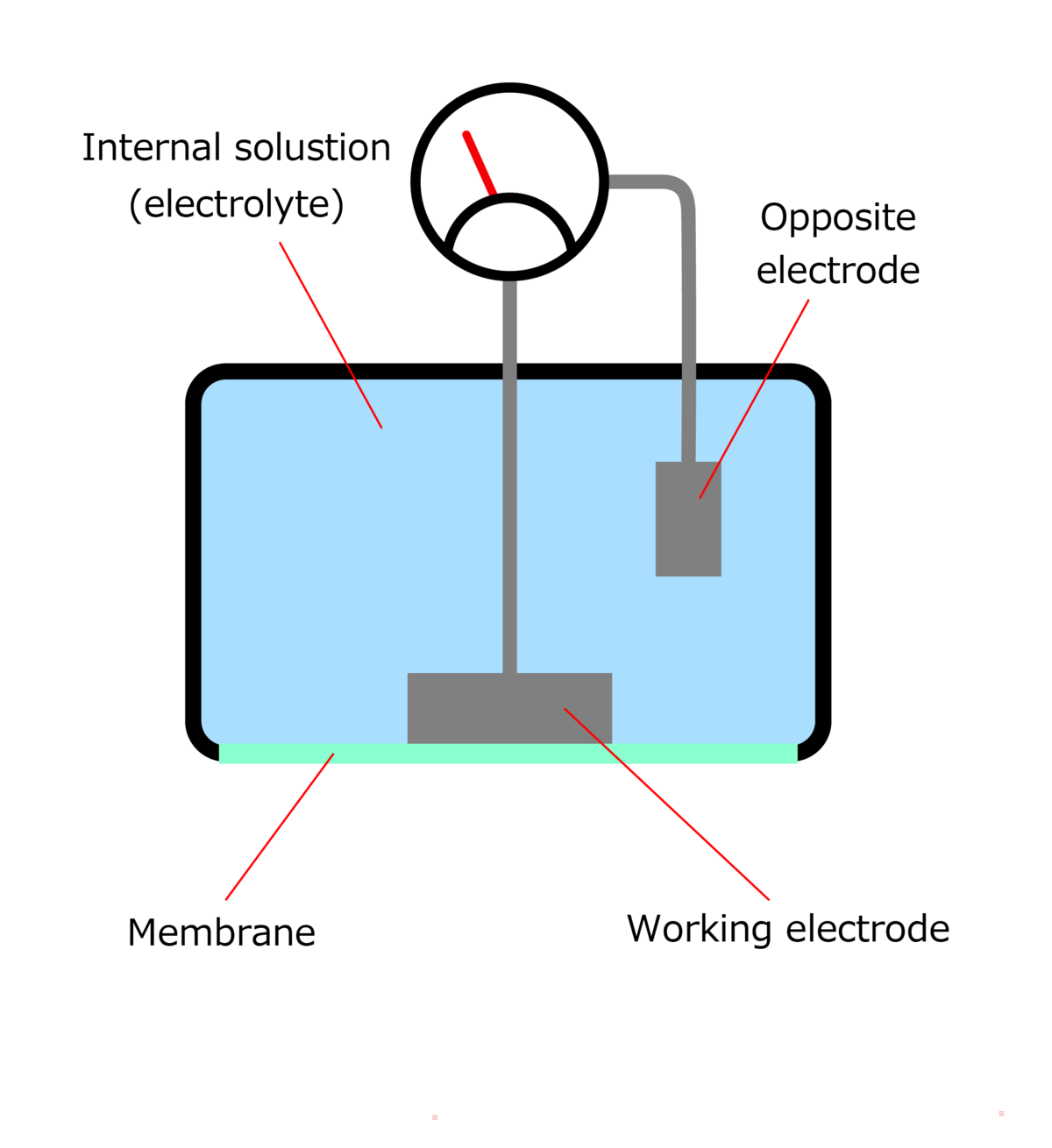
Structure of a membrane (galvanic)-type DO meter
The Galvanic cell method is a method used for measuring dissolved oxygen (DO). This method is an electrochemical technique for measuring oxygen concentration. It uses a membrane that is permeable to oxygen and reduces the oxygen with an electrode to measure DO concentration in a sample based on the current value.
A galvanic cell-type DO electrode is made up of a pair of electrodes made from different materials, a membrane, and internal solution. Oxygen contained in the sample is passed through a membrane with excellent oxygen permeability and absorbed into the internal liquid. A reduction reaction at the working electrode is caused for the absorbed oxygen, producing a current between electrodes. This current is proportional to the DO concentration in the sample, so DO concentration is calculated with an external ammeter.
Structure of a membrane (galvanic)-type DO meter