

Six sites in Inukami River were selected and measured with LAQUA WQ-330 handheld meter and sensors for pH, conductivity, dissolved oxygen, and temperature to determine the water quality. Waters in spring area, which originate from underground, were found with lower pH, DO, conductivity, and temperature compared to waters from mainstream and downstream.

The Inukami River in Shiga Prefecture, Japan is 27.1-km long and flows from the Suzuka Mountains to Lake Biwa. It is known not only for its clean water, but also for its underground flow and numerous springs in its riverbed. In order to preserve the Inukami River water, it is essential to determine the water quality and understand the effect of its ecosystem and surrounding areas.
Six sites in Inukami River were selected for pH, conductivity (EC), dissolved oxygen (DO) and temperature measurements (See Figure 1). These four water quality parameters have important interactions between each of them. For example, conductivity measurement is typically conducted in checking the cleanliness of water. Conductivity increases as temperature and dissolved charged ions (e.g. Na+, Cl-, NO3-) from inorganic substances (e.g., minerals, salts) in water increases. pH is also affected by temperature and minerals dissolved in water. Unlike conductivity, dissolved oxygen concentration decreases as water salinity and temperature increases.
The new LAQUA WQ-330 triple-channel multi-parameter handheld meter was used to simultaneously measure the pH, conductivity, dissolved oxygen, and temperature of Inukami River. Prior to river water measurements, the 300PH-2 / 300-P-C pH sensor, 300-C-2 / 300-4C-C 4-cell conductivity sensor, and 300-D-2 optical DO sensor connected to the meter were calibrated with NIST pH buffers (pH 4.01, 6.86, and 9.18), 84µS/cm conductivity standard, and ambient air, respectively.

As shown in Table 1, the water at confluence point of Inukami River and Ota River ② has higher temperature, pH, conductivity, and dissolved oxygen compared to the waters before ① and after ③ the confluence point. As the tributary Ota River is situated along residential area, it is believed that human activities in the area may have contributed to the slightly elevated water temperature and conductivity obtained at the confluence point. Dissolved inorganic substances (e.g., minerals, salts) coming from sewage may pollute water. The conductivity value of underground flow before the confluence point is affected by geological conditions.
Dissolved oxygen (DO) is free oxygen present in water from atmosphere and photosynthetic production by aquatic plants and algae. There is more dissolved oxygen in cold, flowing water with many rocks or obstacles, and moderate amount of aquatic plants and algae but generally less at the bottom of river. Although the water at confluence point ② is warmer than ① and ③, higher DO was observed which could be due to the merging water flows, aquatic plants, algae, and rocks in the area.
Spring water ⑤ is pumped out from underground and utilized as drinking water in the residential area. The water was found having lower temperature, pH, conductivity, and dissolved oxygen than mainstream water. The riverbed water ④ was found having close results to the spring water ⑤. Due to numerous springs in the riverbed, it is believed that the measured riverbed water ④ originated from the springs.
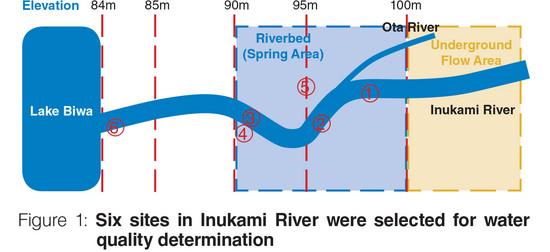
The temperature of water in river is normally affected by the ambient temperature and changes with the seasons. On the other hand, spring water temperature is almost stable since it is not affected by the ambient temperature. Groundwater tends to maintain a relatively long-term average temperature of its aquifer so flow from a spring may be cooler than a summer day but remain unfrozen in the winter.1 This explains the temperature of spring water ⑤ being lower than mainstream water. In stagnant area of a river, sunlight may raise the temperature. This makes the temperature of riverbed water ④ slightly higher than spring water ⑤.
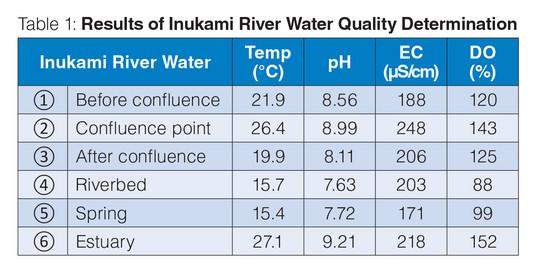
Many springs have naturally low oxygen due to the length of time the water has been underground where no photosynthesis can occur. After leaving the spring vent, gas exchange with atmosphere and photosynthesis from aquatic plants and algae increases the oxygen level as the water flows down the spring run.2 The DO results in spring and riverbed waters were lower than mainstream and estuary waters.
The pH of groundwater is affected by the rocks and chemicals beneath the ground, as well as the quality of the water being recharged or percolating down into the aquifer from the surface. In some places, limestone in aquifer is made mostly of calcium carbonate (CaCO3). Acidic water will react with the carbonate in the limestone, dissolving the limestone, and neutralizing the acid to a pH around 7.3 This could be the reason for the pH values obtained in ④ and ⑤.
Water in estuary ⑥ gently flows around while getting good exposure to sunlight. The measured water pH, DO, and temperature in estuary were the highest results obtained. An area with good exposure to sunlight and water is a conducive environment for aquatic plants to grow. Plants take up carbon dioxide for photosynthesis and release oxygen. This could be the reason for high DO.
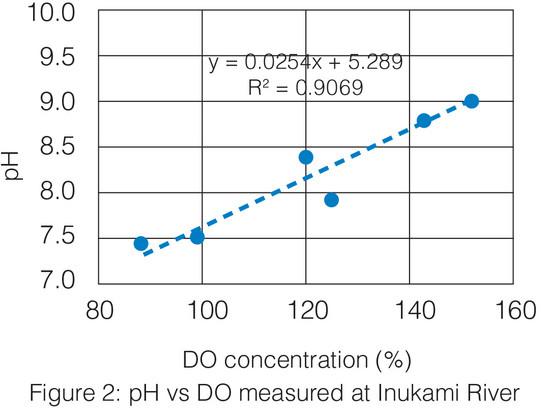
The aquatic plants growing in the water also play a role in affecting the pH. During the day, plants photosynthesize producing both oxygen and bicarbonate ions which shift the pH upward. At night, those same plants respire and release carbon dioxide which lowers the pH.3 This could be the reason for high pH in mainstream and estuary waters at the time of measurement.
Simultaneous pH, conductivity, DO, and temperature measurements provided data that help us understand the Inukami river condition and its water quality. In addition, the measurement results for the different sites selected were as expected considering the factors such as locations, surroundings, and aquatic plants in the Inukami river and also proven that LAQUA WQ-330 is suitable for river water measurements.
12 November 2019, Rev. 0
Handheld Water Quality Meters
Do you have any questions or requests? Use this form to contact our specialists.
HORIBA Advanced Techno, Co., Ltd.
31, Miyanonishi-cho, Kisshoin
Minami-ku Kyoto 601-8306 Japan
Tel: +(81) 75 321 7184
Fax: +(81) 75 321 7291
HORIBA Instruments (Singapore) Pte Ltd.
83 Science Park Drive, #02-02A,
The Curie 118258 Singapore
Tel: +65 6 908 9660
Fax: +65 6 745 8155
Mail: laqua(at)horiba.com
HORIBA UK Limited Northampton Office
Kyoto Close,Summerhouse Road,
Moulton Park
Northampton NN3 6FL UK
Tel: +(44) 1604 542 600
Fax: +(44) 1604 542 699
Mail: waterquality(at)horiba.com
HORIBA Instruments Incorporated Head Office
9755 Research Drive
Irvine California 92618 USA
Tel: +1 800 446 7422
Fax: +1 949 468 1790
Mail: labinfo(at)horiba.com
You might also like to know...
