The activity is a concentration of ions in the solution that is corrected using a coefficient called the activity coefficient, and the relationship between them can be expressed with the following relational expression:
Activity (a) = Activity coefficient (y) • Ion concentration (C)
The following table shows the relationship between the ion concentration and the activity coefficient.
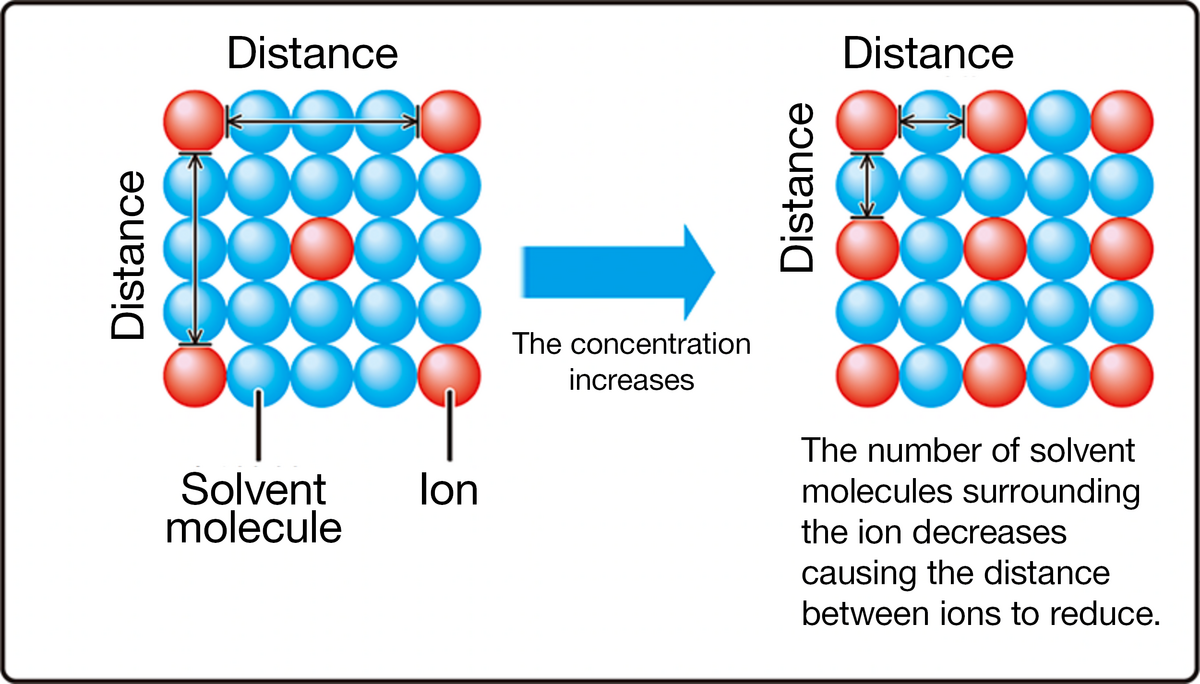
From this table, you can see that when the ion concentration becomes lower than 1 x 10-3 mol/L, the activity coefficient becomes a value close to 1, and as it becomes higher than 1 x 10-3 mol/L, the activity coefficient becomes lower than 1.
If the activity coefficient becomes lower than 1 (activity < ion concentration), an error occurs in the potential measurement. As ISA is used to reduce this error. An ISA is generally selected so that it does not react with the ions to be measured and does not affect the measured potential.
The measurment error can be reduced because adding the ISA enables to maintain the ion strength constant and to adjust the activity coefficients of the sample solution and the standard solution almost at the same value.
(Example) Activity coefficient for monovalent ion concentration
| Ion concentration(C) mol/L | Activity coefficient(γ) |
|---|
| 1×10-5 | 0.998 |
| 1×10-4 | 0.988 |
| 1×10-3 | 0.961 |
| 1×10-2 | 0.901 |
| 1×10-1 | 0.751 |