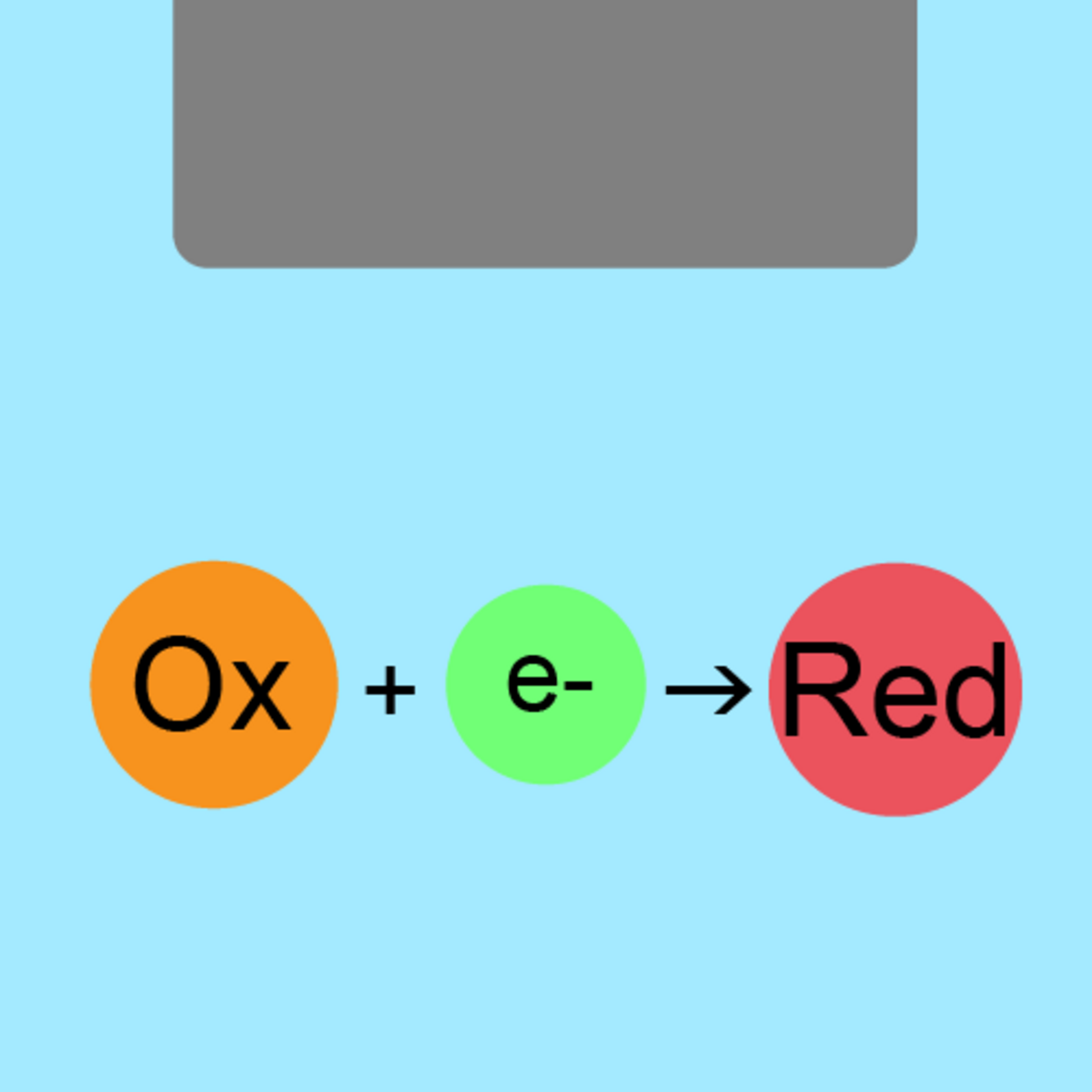
Schematic diagram of oxidation-reduction potential near a metal electrode
Oxidation-reduction potential (ORP) is an indicator showing whether an oxidation reaction or reduction reaction is more likely to occur in a sample. It can be measured by immersing a working electrode with a metal tip (platinum or gold) and a reference electrode into a sample solution. At the metal electrode surface of the working electrode, the sample's intrinsic redox potential can be detected. In addition, the reference electrode demonstrates a constant potential for any sample due to the gradual release of an internal solution (KCl) into the solution. Oxidation-reduction potential can be calculated from the difference in the potential exhibited by the working electrode and the potential exhibited by the reference electrode.
Schematic diagram of oxidation-reduction potential near a metal electrode