The Voltammetry method is a technique for calculating the concentration of substances in a solution by repeatedly applying positive and negative voltages to an electrode placed in the solution and detecting the current flowing at the counter electrode due to reactions with substances in the solution.
When a negative voltage is applied to the working electrode, hypochlorous acid in the solution is electrolytically concentrated onto the working electrode. When switching the applied voltage to positive, hypochlorous acid on the working electrode departs, and the current generated during this process is detected at the counter electrode to measure the hypochlorous acid concentration. (Diagram 1)
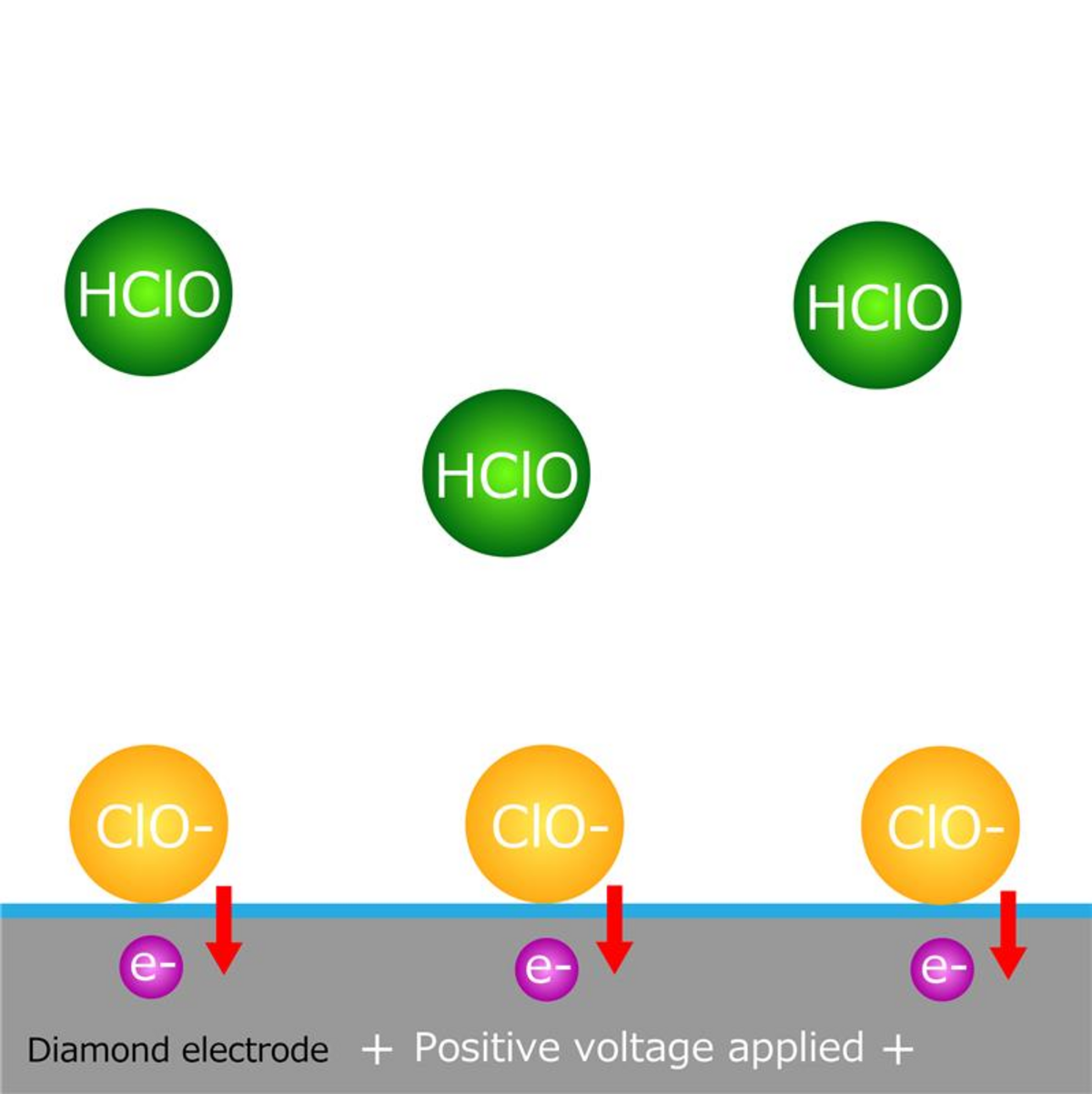
When a positive voltage is applied, hypochlorite ions in the solution are electrolytically concentrated at the working electrode. When switching the applied voltage back to negative, hypochlorite ions depart from the working electrode. By detecting the resulting current at the counter electrode, the concentration of hypochlorite ions is measured. (Diagram 2)
Diagram 2
HORIBA utilizes boron-doped diamond electrodes, which incorporate boron into the diamond electrode to impart conductivity. This enables us to expand the measurement range compared to conventional metal electrodes and achieve electrochemical cleaning through powerful electrolytic polishing.
*The boron-doped diamond electrode is based on the findings of joint research with Prof. Yasuaki Einaga of Keio University.