Malaria is a major global infectious disease, endemic to tropical and subtropical regions worldwide. Despite sustained international efforts aimed at its control and eventual eradication, malaria remains a significant public health burden, causing over 600,000 deaths annually, the majority of which are children.
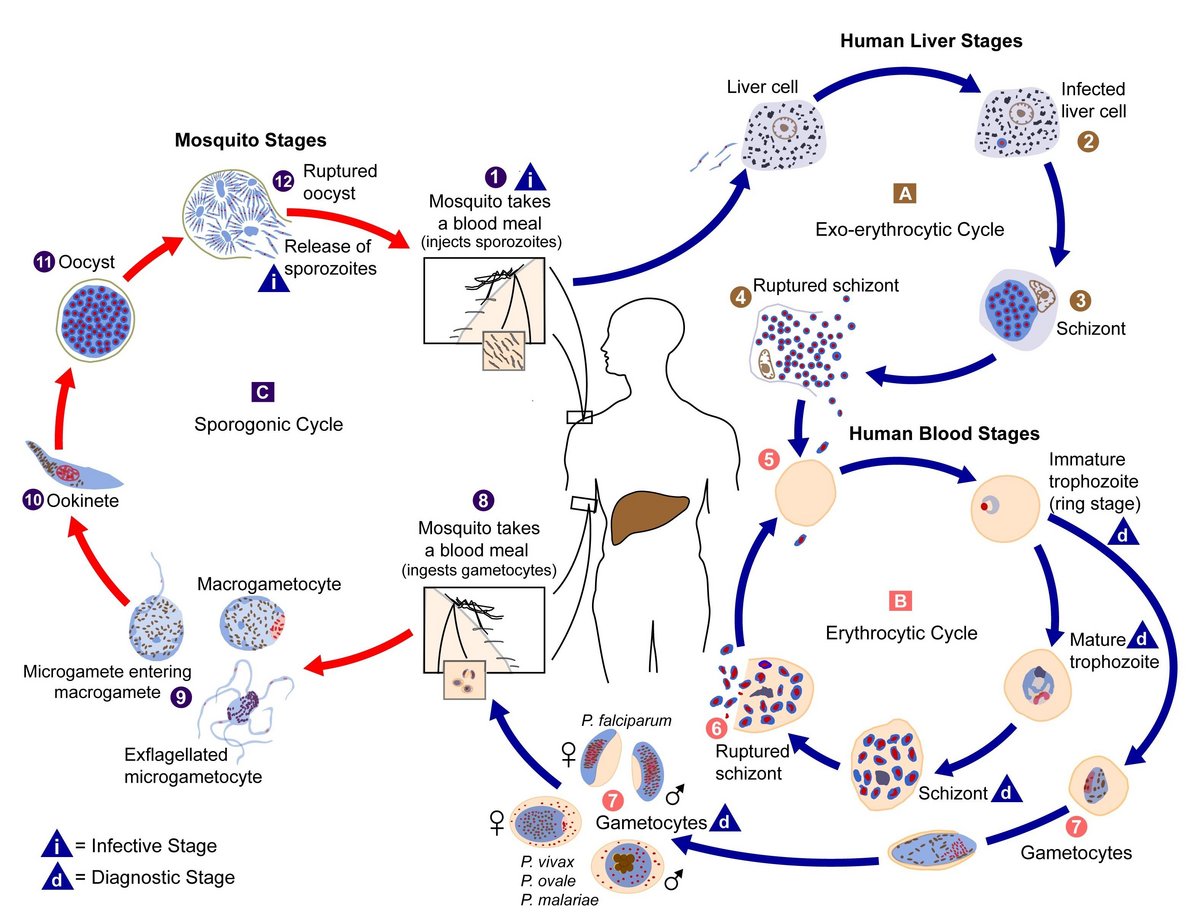
Malaria is transmitted to humans through the bite of infected female Anopheles mosquitoes. During feeding, the mosquito injects Plasmodium parasites into the bloodstream in the form of sporozoites present in its saliva, which quickly migrate to the liver. There, they invade hepatocytes and undergo maturation and asexual replication. This hepatic produces thousands of merozoites, which are released into the bloodstream after rupture of infected liver cells. The merozoites then infect red blood cells, initiating the symptomatic blood stage of the disease. (1)
Five Plasmodium species are known to cause malaria in humans: Plasmodium falciparum, Plasmodium vivax, Plasmodium ovale, Plasmodium malariae, and Plasmodium knowlesi.
Malaria (Plasmodium spp.)
Dengue is another mosquito-borne disease but differs fundamentally from malaria in that it is caused by a virus rather than a parasite. Dengue virus, an RNA virus from the Flaviviridae family, is transmitted primarily by female mosquitoes of the Aedes genus, especially Aedes aegypti. Dengue is endemic in over 120 countries, many of which also experience malaria transmission. Globally, an estimated 390 million dengue infections occur each year, with approximately 500,000 cases requiring hospitalization and around 40,000 deaths. (2)
Despite advances in vector control, diagnostic screening and effective treatments, malaria continues to have a devastating impact on global health.
P.Falciparum - Trophozoites and crescent shaped gametocyte
P.Vivax - Gametocyte (L) and trophozoite (R)
The World Health Organization's Global Technical Strategy for Malaria (2016–2030) aims to reduce malaria cases and deaths by 90% by 2030. (3) Key solutions include mosquito control, improved treatments and prevention, and robust screening and diagnosis. Economically challenged malaria-endemic regions urgently need accessible, reliable diagnostics.
Malaria is typically diagnosed by manual examination of blood smears - either thick or thin - that are stained with Romanowski techniques at a pH of 7.2. Rapid tests and PCR methods are also used. Most symptomatic patients receive a complete blood count (CBC). Malaria symptoms can mimic those of other infections, such as dengue, which is often prevalent in the same regions.
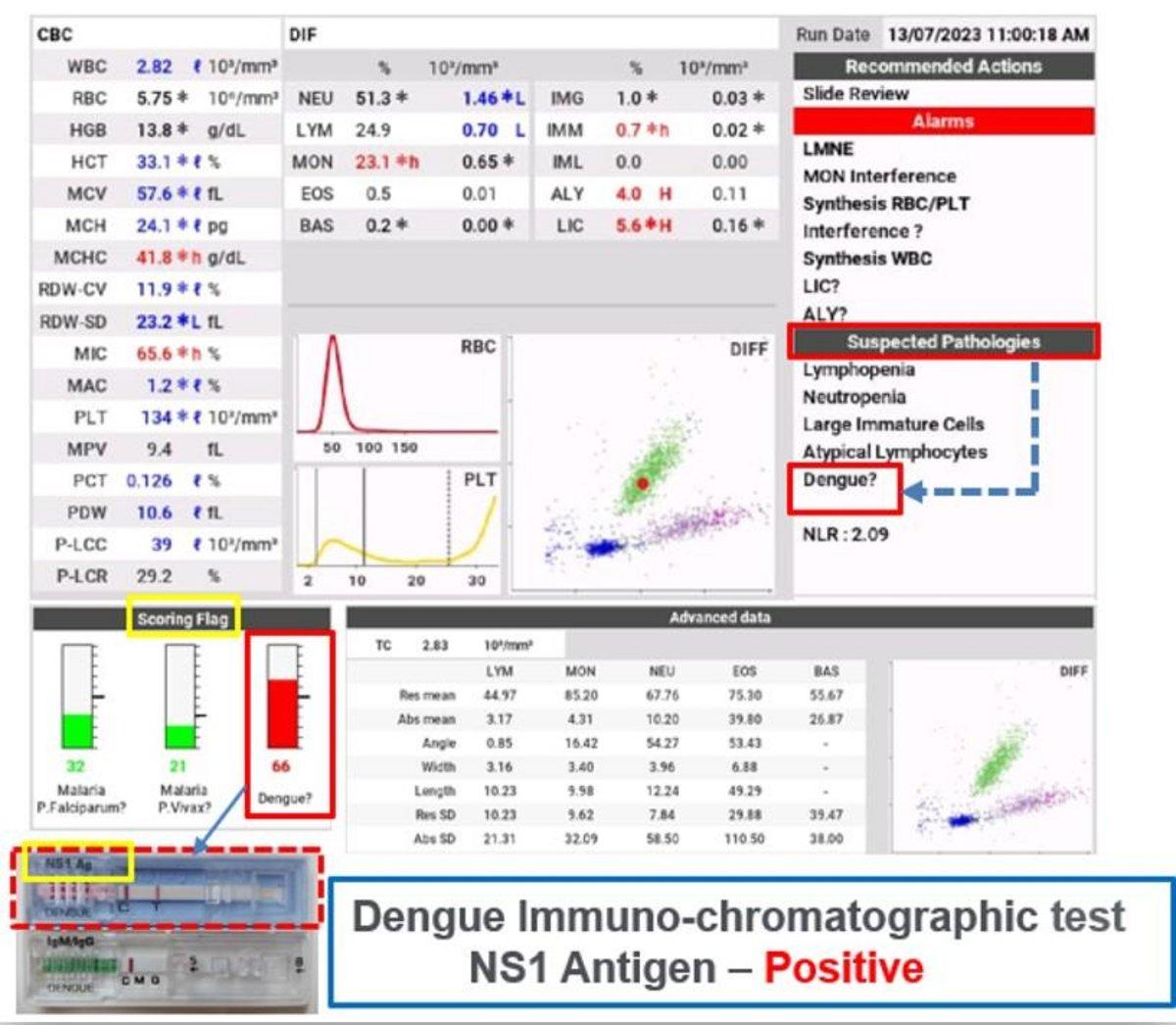
HORIBA has developed infectious screening indicators based on complete blood count data, incorporating advanced algorithms and analysis of thousands of measurement points. (4) The clinical efficacy of these indicators has been evaluated through on-site validation studies for malaria (including Plasmodium vivax and Plasmodium falciparum) and dengue, demonstrating high sensitivity and specificity in clinical settings.
Advanced statistical algorithms enable hematology analyzers to rapidly screen for diseases such as malaria and dengue within minutes, alongside standard CBC reports. The HORIBA 6-part hematology analyzers, Yumizen H550 (autoloader), Yumizen H500 CT (closed tube) and Yumizen H500 OT (open tube), can indicate possible cases of dengue and malaria without requiring additional reagents.
These instruments employ advanced statistical algorithms to differentiate between Plasmodium vivax and Plasmodium falciparum using raw data generated from the device. The infection suspicion notifications act as screening flags to support early identification of suspected malaria and dengue cases.
Flags are integrated with complete blood count analysis (CBC or DIFF mode) and do not require specialized reagents or sampling methods. The algorithms are designed to detect three specific conditions:
The flagging messages are displayed, can be printed, and may be transmitted to a laboratory information system (LIS) or middleware for further review. Flagging scores are presented to indicate the degree of suspicion, helping users assess the reliability of the flagged results.
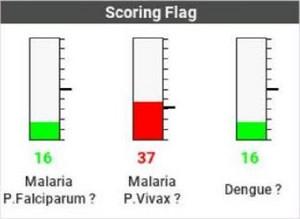
Scoring flags are displayed on the expert data screen on the instrument for further information:
The Yumizen H550 range uses advanced statistical algorithms to develop infectious screening flags from data mining, leveraging bioinformatics techniques for disease biomarkers.
These instruments report 37 hematology parameters, but their raw data are much more detailed, especially regarding resistive and optical cell measurements and scattergram patterns.
The advanced algorithm analyzes this complex information to develop predictive software, refined with clinical cohorts to establish flagging thresholds.
Vivax predictor (left) and dengue predictor (right)
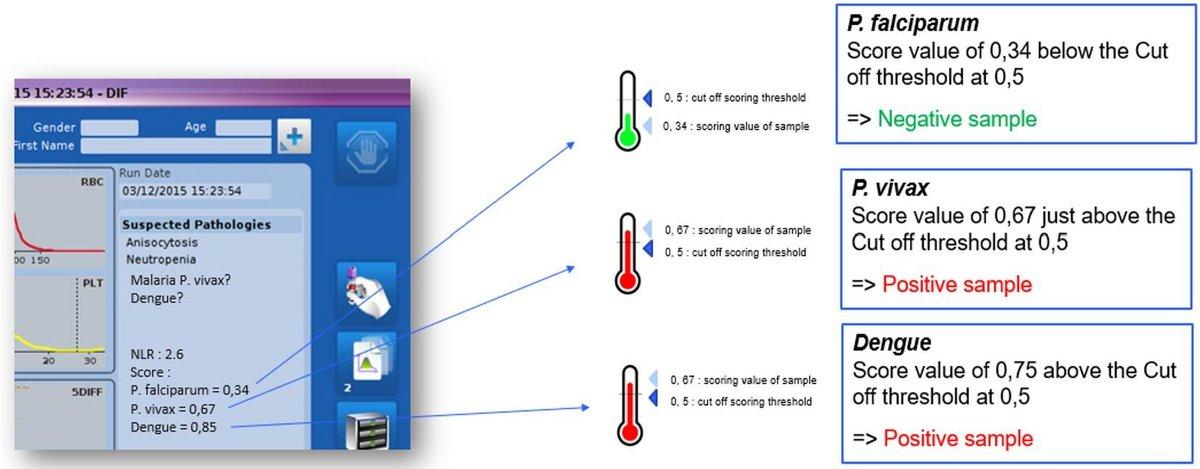
Predictors generate a score representing the likelihood of a specific disease. By adjusting the threshold, the alarm system can be optimized to detect most cases while limiting false positives.
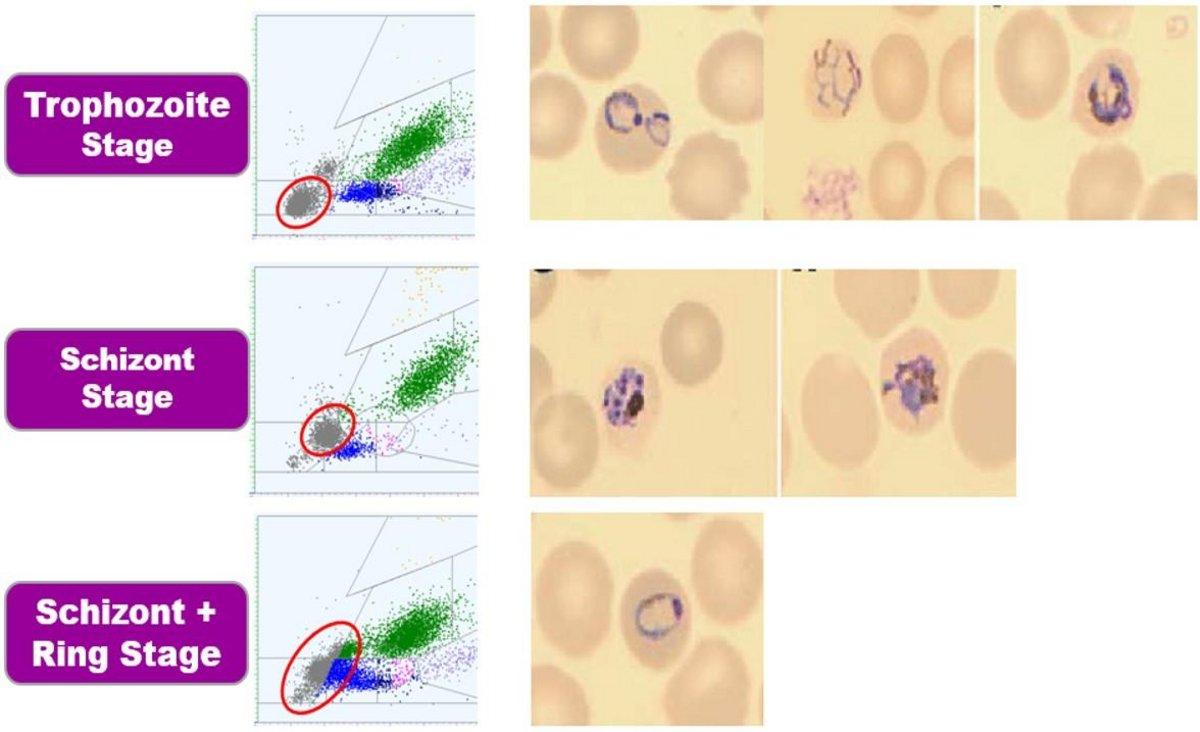
Although the patterns that trigger flags are not always visible on instrument graphics, infected red blood cells (RBCs) are sometimes identifiable on the WBC differential scattergram, appearing to the left of the lymphocyte cell population. In malaria caused by the vivax species, visually identifying infected RBCs can confirm a positive sample.
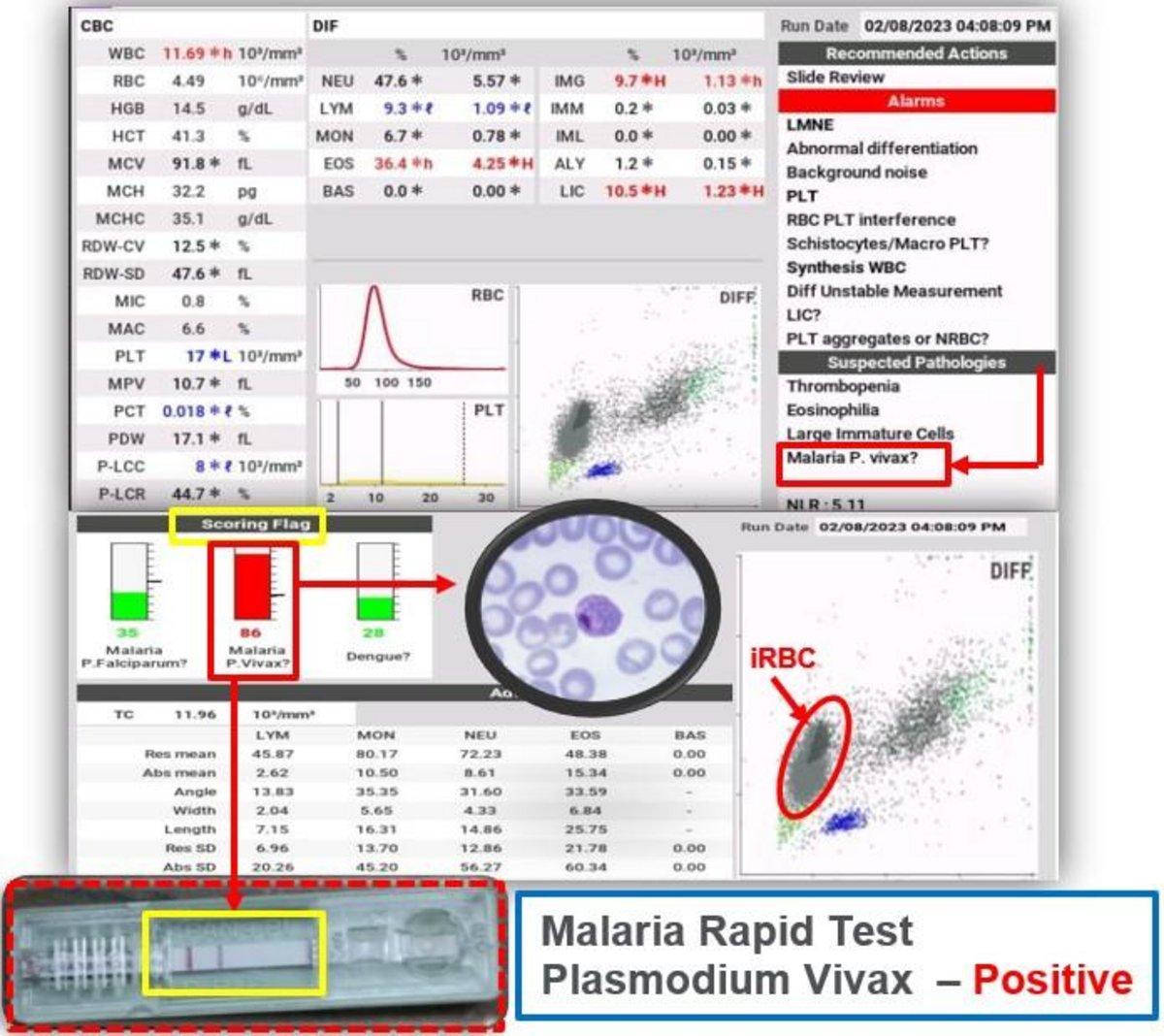
Malaria infection, especially Plasmodium vivax, can interfere with total WBC counts because parasitized RBCs affect measurements. HORIBA Yumizen H550 and Yumizen H500 hematology analyzers automatically correct WBC counts by excluding infected RBCs, whereas most other analyzers may show interference in total WBC counts.
Malaria infected RBCs in Yumizen H550 WBC scattergram
Malaria and dengue screening flags are also available on the latest Yumizen H500 models with onboard CRP or ESR testing, including Yumizen H500 CRP, Yumizen H550E, and Yumizen H500E. These systems integrate automated infectious disease screening with inflammatory biomarker assessment in a single workflow.
The simultaneous availability of malaria and dengue suspicion flags alongside ESR or CRP measurements provides complementary clinical information at initial testing. In suspected cases, CRP helps differentiate viral from bacterial co-infection and supports assessment of disease severity or complications, while ESR offers a broader indicator of systemic inflammation and disease progression. This integrated hematology–inflammation approach supports early clinical triage and more informed patient management, particularly in endemic or resource‑limited settings where rapid, consolidated diagnostics are critical.
► Malaria infection
In this patient, a distinct population was observed on the left side of the matrix. The algorithm produced a highly positive indicator for Plasmodium vivax, which was confirmed by blood smear examination and a Malaria Rapid Test.
► Dengue infection
In this case, a patient with fever showed mild neutropenia, relative monocytosis, and mild thrombocytopenia. The instrument flagged dengue, which was confirmed through antigenic testing.
Discussion
Numerous studies worldwide have evaluated the sensitivity and specificity of automated infectious flags, suggesting these tools could effectively screen for malaria and dengue in clinical environments. (4)
A pilot study conducted across four regions in India measured flag sensitivity: 90% for P. vivax and 100% for P. falciparum, with specificity rates of 95.01% and 99.59%, respectively. (5)
Thresholds can be modified based on population data and specific needs; a higher cut-off increases specificity. These alarms may indicate various infections. Because instruments provide results within minutes, early screening can reduce testing costs.
Note: Clinical correlation and additional testing are necessary to confirm diagnoses.
Conclusions
Yumizen H550 and Yumizen H500 hematology analyzers provide accurate, automated, and rapid malaria screening. In prospective studies, their flags performed comparably to microscopy.
HORIBA Yumizen H550 and Yumizen H500 hematology analyzers offer cost-effective malaria screening in resource-limited, endemic regions, supporting further diagnosis and prevention of drug resistance.
For more details, please do visit https://www.horiba.com/int/medical/academy/technology/malaria-dengue-screening
Beyond screening and diagnostic support, effective malaria management also requires consideration of treatment safety, particularly in regions where glucose‑6‑phosphate dehydrogenase (G6PD) deficiency is common. G6PD deficiency is the most prevalent enzymatic blood disorder worldwide and is highly prevalent in malaria‑endemic regions. While many individuals with G6PD deficiency are asymptomatic, certain antimalarial drugs - particularly primaquine or tafenoquine - can trigger acute hemolytic episodes.
For this reason, international guidelines recommend assessment of G6PD enzymatic activity before starting treatment, particularly for Plasmodium vivax and Plasmodium ovale infections, where relapse‑preventing therapies are indicated. Quantitative G6PD testing enables accurate determination of enzyme activity across the clinical spectrum, including intermediate phenotypes that qualitative or rapid tests may miss.
HORIBA provides quantitative G6PD test reagents for precise evaluation of G6PD activity as part of the malaria care pathway. (6)
When integrated alongside hematology‑based malaria screening, G6PD testing supports informed treatment decisions, helping clinicians balance therapeutic efficacy with patient safety in line with current international recommendations.
Hematology Analyzer
Hematology Analyzer
Hematology and ESR Analyzer
Tiene alguna pregunta o solicitud? Utilice este formulario para ponerse en contacto con nuestros especialistas.