If specific ions exist in the sample solution, the ion selective membrane of the ion selective electrode generates a potential that corresponds to the concentration of those ions. The generated potential is measured as the potential difference on the basis of the reference electrode using an ion meter or meter with an ion measurement function.
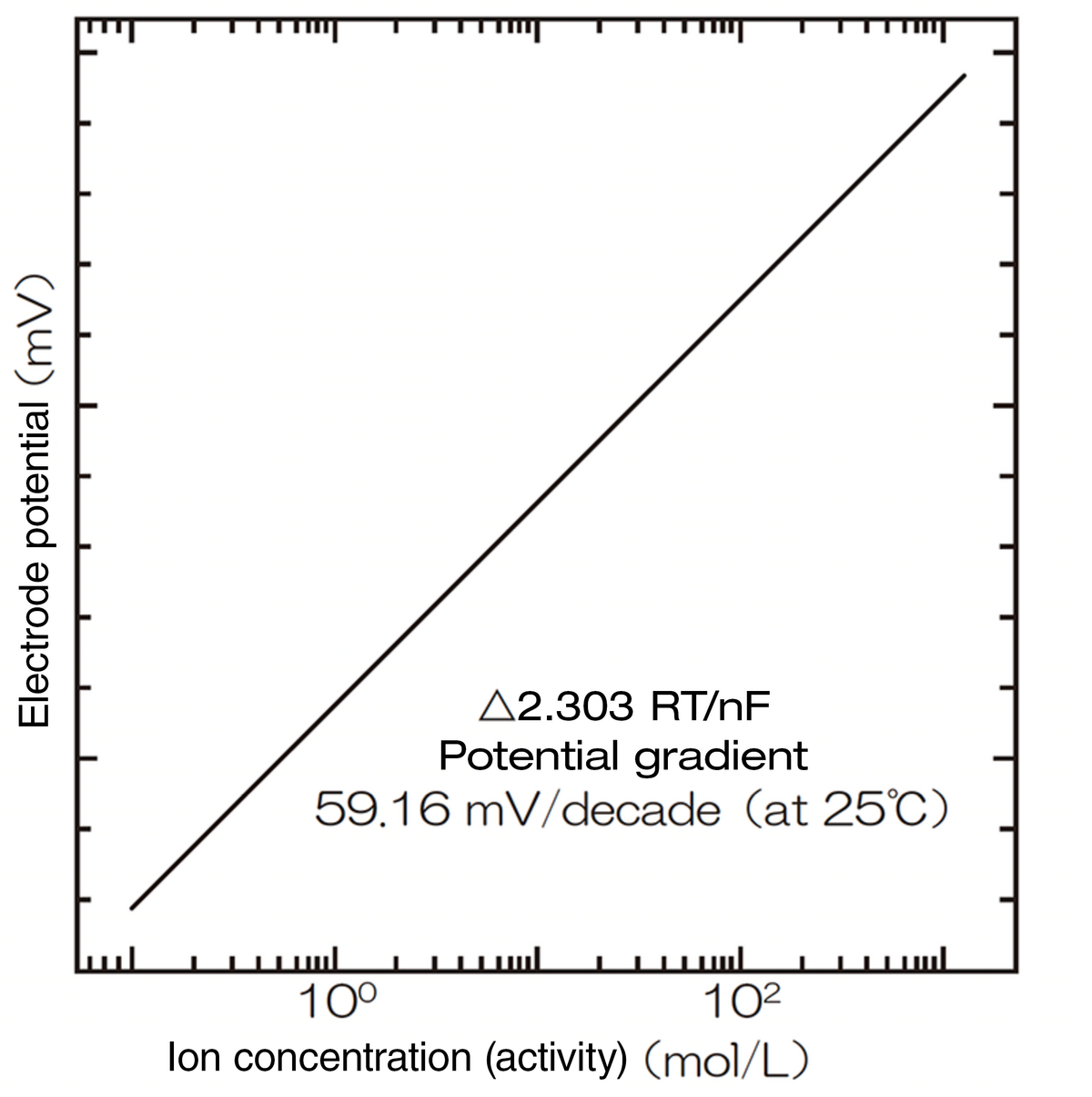
The measured potential difference and the logarithm of the concentration (activity) of the ions to be measured in the sample solution are in a proportional relationship which can be represented by the following equation:
E = E0 + (2.303 RT/nF) logyC
E: Measured electrode potential (V)
E0: Reference potential depending on the ion selective electrode (V)
(The reference potential of the reference electrode and the potential difference of the liquid junction are included.)
F: Faraday constant (96485 C mol-1)
R: Gas constant (8.314 JK-1 mol-1)
T: Absolute temperature (K)
n: Ion charge number
y: Activity coefficient
C: Ion concentration
This equation is called the Nemst equation, and is the base of ion concentration measurement using an ion selective electrode.
(2.303 RT/nF) is the change of the electrode potential generated when the ion concentration (activity) is changed by a factor of ten times, and is referred to as the sensitivity, potential gradient, slope, or Nemst coefficient.
According to the above equation, if the potential gradient is determined by the calibration using the standard solutions, the concentration (activity) A of the targeted ions can be measured by obtaining electrode potential E of the ion selective electrode in the sample solution.