

Pharmaceutical aerosols including metered-dose inhalers (MDIs) and dry powder inhalers (DPIs) are devices that deliver a specific quantity of drug to the lungs. The particle size distribution and shape of the delivered dose is more critical for inhalation aerosols than for most other conventional drug products because these factors greatly influence the deposition profile in the lungs of the patient. The optimum aerodynamic particle size distribution for most inhalation aerosols has generally been recognized as being in the range of 1-5 µm.
Although the compendial tests for pharmaceutical aerosols are based on cascade impactors, microscopic analysis can provide valuable information on particle size and shape distribution of the drug particles such as:
The MDI device consists of a canister, and actuator, and sometimes a spacer. The canister itself consists of a metering dose valve with an actuating stem. The formulation resides within the canister and is made up of the drug, a liquefied gas propellant, and often stabilizing excipients. Actuation of the device releases a single metered dose of liquid propellant that contains the medication. The volatile propellant breaks up into droplets which then evaporate, creating an aerosol containing micronized drug that is inhaled into the lungs.
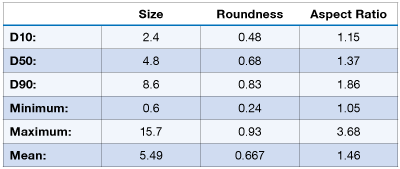
The dose delivered by an MDI can be analyzed using a microscope, or preferable an automated image analyzer. A brochodiator MDI was examined using the PSA300 image analysis system. The particle size and shape distribution is shown below:
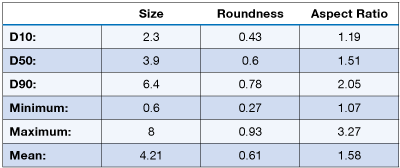
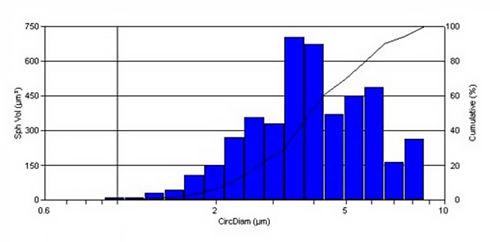
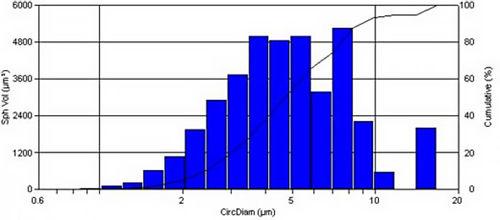
DPIs are an alternative to the aerosol based inhalers commonly MDIs, that deliver a powder dosage form to the lungs. Most DPIs include an active ingredient and one or more excipient to aid powder dispersion and flow. The powder dose from a DPI can be analyzed on the PSA300 image analysis system.

Results from a bronchodilator DPI containing salmeterol and fluticasone are shown below:
Static Image Analysis System Particle Size
Laser Scattering Particle Size Distribution Analyzer
Nanoparticle Analyzer
Do you have any questions or requests? Use this form to contact our specialists.