
Modifications on the SPRi-Biochip surface (at about 200 nm), such as an interaction between ligands and analytes, induce a change of resonance conditions, which can be measured.
Surface plasmon resonance (SPR) is an optical detection process that can occur when polarized light hits a prism covered by a thin metal layer. Under certain conditions (wavelength, polarization and incidence angle), free electrons at the metallic surface of the SPRi-Biochip absorb incident light photons and convert them into surface plasmon waves. These waves are penetrating the medium above the surface of about 100 to 200 nm. At a specific incidence angle (called resonance angle), the plasmon waves resonate with the incident light, resulting in a dip of reflectivity. The plasmon curves show the variations of the reflected light collected on the camera versus the incidence angle. The position of the resonance angle is very specific to the local environment of the SPRi-Biochip surface.
Modifications on the SPRi-Biochip surface (at about 200 nm), such as an interaction between ligands and analytes, induce a change of resonance conditions, which can be measured.
A plasmon is the physical phenomenon characterizing plasma oscillation. More precisely, in physics, in the same manner as photons are the quantification of light waves, a plasmon is a quantum of plasma oscillation. The plasmon is the quasiparticle resulting from the quantification of plasma oscillations. Thus, plasmons are collective oscillations of the free electrons gas density, often at optical frequencies. They can couple with a photon to create a third quasiparticle called a plasma polariton.
In surface plasmons, instead of having collective oscillations of a gas, we have a collective oscillation of the conduction electrons of a metal. More precisely, surface plasmons are plasmons that are confined to surfaces and that interact strongly with light, resulting in a surface plasmon polariton (or surface plasmon wave).
The flow cell image retrieved by the software corresponds to the real time image of the SPRi-Biochip surface. The difference image shows interacting spots when the analyte solution is injected.
Imaging allows simultaneous monitoring of the resonance conditions on the surface of the SPRi-Biochip. If the SPRi- Biochip contains different immobilized molecules (array format), potential interactions on those different molecules (also called ligands or spots) are monitored in parallel, thanks to the use of a camera. This property (simultaneous measurement on all the elements of the microarray) is also often referred to as multiplexing.The imaging feature of our devices enables the multiplex measurement of up to several hundred interactions.
The flow cell image retrieved by the software corresponds to the real time image of the SPRi-Biochip surface. The difference image shows interacting spots when the analyte solution is injected.
Kinetic interaction curve: (A) latency time, (B) association, (C) saturation, (D) dissociation, (E) regeneration.
When the sample solution is injected, the analyte must reach the flow cell to interact with the ligand. This period of time when the analyte is not yet in the flow cell is called the latency time (A).
Then the sample is injected, analytes go through the flow cell and they interact with the ligands on the surface. This step is called the association phase (B). A plateau is reached (C) at the end of the association and this can be explained by molecular statuses (equilibrium, saturation or volume injected finished). When no more analytes go through the flow cell, the solution of analytes is replaced by the running buffer, and analytes dissociate from the immobilized ligands, it is the dissociation phase (D).
Some interactions have a high affinity and some analytes may still bind to the ligands after the dissociation step. In this case, a regeneration solution is injected to remove all the remaining analytes. This is the regeneration step (E). A new sample solution can then be injected.
Kinetics parameters
ka is the association rate constant (M-1.s-1), in some cases, it is also called kasskd is the dissociation rate constant (s-1), in some cases, it is also called kdiss.
SPR kinetics data (interactions) can be modeled simply in order to evaluate the affinity between 2 molecules. The model generally used assumes that the reactions involve one ligand (L) and one analyte (A) which interact with a 1:1 stoichiometry:
![]()
This model gives the following formulae for our interaction kinetic curves, one for the association response (Ra) and one for the dissociation response (Rd), the response being in our case, the reflectivity variation:
Ra(t)=Req•(1-exp[-kobs•(t-t0)]) (association)
Rd(t)=Rd(t1) • exp[-koff•(t-t1)] (dissociation)
with kobs = kass • Ci+kdiss, the observed on-rate constant of the response during association, Ci the injected analyte concentration and koff = kdiss, the off-rate constant of the response during dissociation, t0 and t1 are indicating respectively the beginning of the association and of the dissociation.
Briefly, the kinetics parameters kass and kdiss are calculated by fitting each part of the interaction (association and dissociation) with a mono-exponential curve, preferentially by using the ScrubberGen or EzFit software (Scrubber-based).
Affinity parameters
KA is the affinity constant (M-1)
KD is the equilibrium constant (M)
Knowing the kinetics parameters, the affinity constant between 2 molecules can be deduced:
KA = 1/KD = ka/kd
The affinity parameters can also be calculated from equilibrium values with a Langmuir fit.
Thermodynamics parameters
ΔG is the free energy change
ΔH is the enthalpy change
ΔS is the entropy change
It can be calculated from the affinity constant (or from the dissociation constant):
ΔG = R • T • ln(KD)
where R is the gas constant, T is absolute temperature (K) and KD is expressed in M.
In order to calculate ΔH and ΔS, the affinity parameters must be determined at several temperatures. ΔG is deduced from these values over a range of temperatures. The non-linear Van’t Hoff equation is fitted to the data to determine ΔH, ΔS:
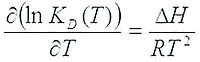
Then, knowing that ΔG = ΔH-T•ΔS, the entropy change can also be evaluated.