

► Morphology Case Study
► Glucose-6-Phosphate Dehydrogenase
► Quiz
► PDF
Patient Details: Male, age 76, attending the dermatology unit
| WBC 11.1* (10^3/mm3) |
| RBC 4.4 (10^6/mm3) |
| HGB 13.6 (g/dL) |
| HCT 41.0 (%) |
| MCV 94.3 (fL) |
| PLT 322 (10^3/mm3) |
Smear Comment: Presence of a population with a lymphomatous appearance, often showing a very dense nucleus (sometimes cleaved), a high N/C ratio, with little cytoplasm. Lymphocyte immunophenotyping results: Identified pathological population of T lymphocytes: CD3 (pos), CD4 (pos), CD7 (neg), CD2 (neg), CD158k (pos).
Combined results in favor of a diagnosis of Sezary lymphoma.
Blood Smear Examination: The blood smear shows a number of large lymphocytes with a cerebriform or convoluted nucleus and scant to moderate basophilic cytoplasm.
Lymphocytes (Normal)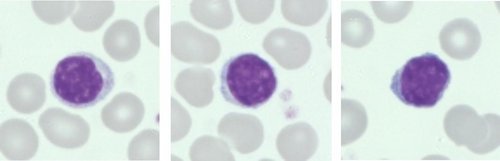
Sezary Cells (Abnormal)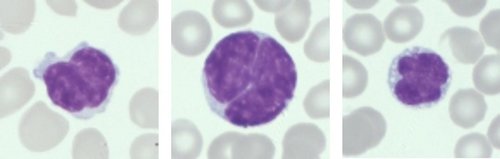
Neutrophils (Normal)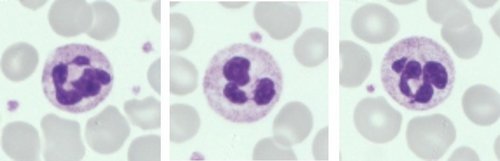
Monocytes (Normal)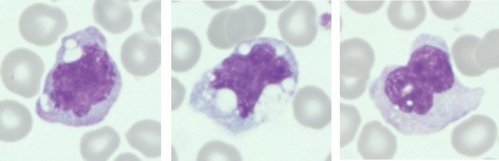
Flow cytometry is crucial for a full and correct diagnosis.
Sezary syndrome is an aggressive T-cell cutaneous lymphoma seen in middle-aged to elderly patients and shows a male predominance. Patients present with erythroderma (redness affecting >80% of the skin), palmar and plantar hyperkeratosis (thickening of the skin on the soles of the feet), intense pruritus (itchy skin), alopecia (hair loss), hepatomegaly, and lymphadenopathy.
Glucose-6-phosphate dehydrogenase or G6PD is an enzyme that protects cells from damage caused by oxidative stress. G6PD is the rate-limiting enzyme of the pentose phosphate pathway (hexose monophosphate shunt). In this metabolic pathway, G6PD supplies reducing energy to red blood cells (RBCs) through NADPH and glutathione (GSSG), which can protect RBCs from oxidative stress which would otherwise damage hemoglobin and red cell membrane. Since RBCs have no mitochondria, the only source of NADPH is from the pentose phosphate pathway, meaning that G6PD levels are essential to the correct functioning of red cells. G6PD is found in all organisms except for Archaea, which are mostly anaerobic organisms. G6PD is expressed in every human cell, indicating its pivotal role in protecting cells from damage (knockout of the G6PD gene is lethal early in embryonic life). G6PD activity decreases as the cell ages, with the most mature cells having approximately one-tenth of the original activity. Reticulocytes have a high level of G6PD activity (a point needed to be noted when investigating patients with high levels of reticulocytes).
The G6PD gene is located on the X chromosome; therefore, males have only one copy of the gene, whereas females have two copies (i.e., it is sex-linked).
Males can be either hemizygous G6PD normal or hemizygous G6PD deficient. Females, on the other hand, can be homozygous G6PD normal, homozygous G6PD deficient (a rare occurrence), or heterozygous for G6PD deficiency. Due to the process of X-linked inactivation or lyonization where one of the X chromosome is silenced in each cell, heterozygous females therefore exhibit differing levels of G6PD activity, ranging from G6PD normal to those levels expected in hemizygous males.
There are approximately 230 G6PD variants with known mutations, most of which give rise to decreased G6PD activity (G6PD deficiency). The degree of G6PD activity is variable among the different mutations. The decrease in G6PD activity is probably due to an increase in molecular instability or decrease in catalytic activity rather than a decrease in production. Due to the nature of inheritance, males can carry either an affected G6PD gene or a normal G6PD gene. The highest frequency of G6PD deficiency is found in sub-Saharan Africa, the Middle East, and parts of Asia.
G6PD Deficiency
G6PD deficiency is the most commonly observed enzyme deficiency in the world with approximately 400 million people affected worldwide. Most people with G6PD deficiency remain asymptomatic throughout their life, however, severe acute hemolytic anemia may present after ingestion of fava beans or by a number of commonly prescribed drugs (e.g., primaquine, rasburicase, dapsone) or even with infection.
Different mutation variants predominate by region: the G6PD A− variant is most common in Africa and the Americas, while the G6PD Mediterranean variant is prevalent in the Middle East and the Mediterranean basin. The prevalence of G6PD deficiency closely overlaps regions where malaria is (or was) endemic, indicating that G6PD deficiency offers a selective advantage by protecting against severe malaria. G6PD deficient cells have higher oxidative stress, which makes it difficult for malaria parasites to survive and replicate; infected deficient cells are also damaged prematurely and removed quickly during passage through the spleen. The downside of this is that common antimalarial drugs like primaquine and tafenoquine trigger oxidative stress which can cause life-threatening hemolytic anemia if given to G6PD deficient patients.
In G6PD deficiency, hemolytic anemia is caused when an offending agent (primaquine or derivatives of fava beans) generates reactive oxygen radicals. G6PD deficient RBCs cannot detoxify these entities due to reduced NADPH and glutathione. For a list of causative agents, see Table 1. Subsequent oxidative damage causes denaturation of hemoglobin, creating Heinz bodies and, subsequently, bite cells (RBCs that appear as if a “bite” has been removed; also called “bitten-out” cells or degmacytes), along with membrane damage and rigidity, splenic removal of damaged red cells, and intravascular and extravascular hemolysis. Patients present with clinical anemia, jaundice, abdominal pain, and sometimes fever. The spleen may be enlarged, and the urine is usually dark (hemoglobinuria). A CBC will show decreased RBC count and hemoglobin; LDH and unconjugated bilirubin will be raised; and haptoglobin (if performed) will be decreased. A reticulocyte count should be performed. A Heinz body preparation can be performed by staining EDTA blood using a supravital stain such as crystal violet or brilliant cresyl blue: incubate for 10–20 minutes, make a thin smear, then view microscopically. Heinz bodies will present as purple stained inclusions near the red cell membrane. A direct Coombs test should be performed to exclude possible autoimmune hemolytic anemia. G6PD activity testing during the acute episode should be viewed with caution as the most deficient cells are being replaced with reticulocytes with a higher G6PD activity, which can give false “normal” results. G6PD activity testing should be assessed after the hemolysis has resolved, preferably 6–8 weeks post episode.
If the anemia is severe, a transfusion may be urgently required, otherwise, only fluid support and analgesics may be needed. If drug-related, removal of the drug is necessary. Note that some drugs have a longer half-life than others (e.g., tafenoquine has a very long half-life of 15–17 days compared to 6 hours for primaquine).
Rare patients present with chronic non-spherocytic hemolytic anemia. The clinical picture is similar to that of hereditary spherocytosis (e.g., jaundice and gallstones), with wide severity ranging from mild to severe enough to require transfusion. Unlike in hereditary spherocytosis, a history of neonatal jaundice (sometimes severe) is the rule.
The World Health Organization (WHO) published a classification of G6PD related to its activity and hemolysis risk.
| Revised WHO Class | Enzyme Activity (% of normal) | Baseline Hemolysis Risk |
| Class A | < 20%a | Chronic hemolysis |
| Class B | < 45% | Acute, trigger‑induced hemolysis |
| Class C | > 60% | No hemolysis |
| Ub | Any | Uncertain clinical significance |
a A variant with <20% activity will be in Class A only if it is associated with chronic hemolytic anemia. If a variant with <20% activity is associated with acute hemolytic anemia induced by fava beans, drugs, or infection, it will be in Class B. If clinical manifestations are unknown, it will be in Class U.
b Temporary assignment for variants for which there is currently insufficient information regarding clinical manifestations.
This classification is used to clarify the drug regime for patients with malaria (e.g., as shown below).
| Feature | Class A | ClassB | Class C |
| G6PD activity | < 20% | 20–60% | > 60% |
| Blood-stage Tx | ✅ ACT /CQ | ✅ ACT /CQ | ✅ ACT /CQ |
| Primaquine (daily) | ❌ | ❌ | ✅ |
| Primaquine (weekly) | ❌ | ✅ (8 weeks) | ❌ |
| Tafenoquine | ❌ | ❌ | ✅ (≥70%) |
| Radical cure | ❌ Not possible | ⚠️ Limited | ✅ Standard |
| Relapse risk | High | Reduced | Minimal |
Note: Chloroquine or ACT (e.g., artemether–lumefantrine) can be used for WHO Class A.
Table 1. Drugs That May Trigger Acute Hemolytic Anemia (AHA) in G6PD-Deficient Patients
| Definite risk of AHA | “Possible” risk of AHA |
| Antimalarial drugs | |
| Dapsone-containing combinations | Chloroquine |
| Pamaquine | Quinidine |
| Primaquine | Quinine |
| Tafenoquine | |
| Other drugs | |
| Ciprofloxacin | Aspirin† |
| Glibenclamide | Menadiol sodium phosphate |
| Methylthioninium chloride | Sulfadiazine |
| Moxifloxacin | Sulfasalazine |
| Nalidixic acid | Sulfonylureas |
| Niridazole | Tolonium chloride |
| Nitrofurantoin | |
| Norfloxacin | |
| Ofloxacin | |
| Phenazopyridine | |
| Rasburicase and pegloticase | |
| Sulfamethoxazole/ cotrimoxazole | |
Measuring G6PD
The recommended reference range for G6PD activity (measured at 37°C) is:
12.1 ± 2.09 U/g Hb
351 ± 60.6 U/1012 RBC
There are two types of G6PD testing: qualitative and quantitative. Qualitative tests (e.g., the fluorescent spot test [FST] or a lateral flow test) can provide a quick decision as to whether a patient is G6PD deficient or not. They are relatively inexpensive and can be performed quickly. Both rely on assessing NADPH production over a defined time period. These tests are adequate for detecting at picking up severe levels of G6PD deficiency but are less reliable in testing females and during a hemolytic crisis. Any patient being deficient should have a quantitative G6PD test performed to confirm the level of deficiency.
Quantitative tests like the Pointe G6PD reagent supplied by HORIBA on its biochemistry platforms measure G6PD concentrations. G6PD catalyzes the reaction glucose-6-phosphate + NADP⁺ → 6-phosphogluconate + NADPH. Therefore, NADPH production is proportional to the concentration of G6PD, and the rate of absorbance change at 340 nm is related to the concentration of G6PD.
A patient presents with low Hb, RBC, normal MCV, but with very low platelet count. The blood smear shows the following red cell abnormality and occasional nucleated red cells. What condition may this be caused by?
a) Microangiopathic Thrombocytopenia in conjunction with Microangiopathic Hemolytic Anemia
b) Iron deficiency
c) Megaloblastic anemia
Answers are revealed two weeks after launch—check your inbox, our socials (LinkedIn / Facebook / X), or the next issue.

The presence of Auer rods in blast cells is most strongly associated with which condition?
a) Chronic lymphocytic leukemia
b) Acute myeloid leukemia
c) Iron deficiency anemia
The Answer: b) Acute myeloid leukemia
Auer rods are needle-like inclusions seen in myeloid blasts and are characteristic of acute myeloid leukemia.

Bibliography
Glucose-6-phosphate dehydrogenase deficiency, blood® 10 SEPTEMBER 2020 | VOLUME 136, NUMBER 1, Lucio Luzzatto, Mwashungi Ally, and Rosario Notaro https://doi.org/10.1182/blood.2019000944
G6PD deficiency and testing. https://www.horiba.com/int/healthcare/products/clinical-chemistry/g6pd-deficiency-and-testing/
Editorial Team
Kelly Duffy, Andrew Fisher, HORIBA UK Limited