

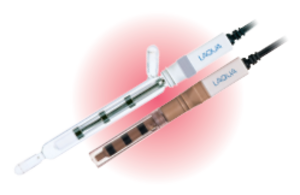
Conductivity cells, also called conductivity electrodes or probes, are constructed with metal electrodes placed at a fixed distance in either glass or plastic body and surrounded by an outer tube. The distance between the electrodes divided by their surface area is known as cell constant. HORIBA offers two-electrode conductivity cells with cell constants expressed in cm-1 and m-1 units.
The cell constant and performance of a conductivity cell may degrade over time due to fouling of electrodes or peeling of their platinum black coating. To maximize the performance of a conductivity cell and extend its life span, proper care and regular maintenance are equally required.

1M Hydrochloric acid (HCl) or Household bleach

Clean water (e.g., tap, distilled or deionized water) in a squirt bottle or beaker

Mild detergent

Soft lint-free tissue
Refer to the safety data sheet (SDS) of any chemical solution that will be used in cleaning and wear the appropriate personal protective equipment (PPE) for safe handling.
1. Cell Type
HORIBA has two types of conductivity cells—submersible and flow-through. The differences between the two conductivity cells are outlined in the table below.
2. Cell Constant
Each type of conductivity cell has four models with 0.1 cm-1 (10 m-1), 1.0 cm-1 (100 m-1) and 10 cm-1 (1000 m-1) cell constants. When selecting a conductivity cell, check the expected conductivity of the sample and then look at the cell constants of conductivity cells. Choose a conductivity cell that covers the expected sample conductivity. The higher the conductivity value of sample, the higher the cell constant required. A conductivity cell with 1.0 cm-1 (100 m-1) cell constant is the most commonly used because it measures low to high conductivity.
3. Body Materials
To avoid compromising the appearance and performance of a conductivity cell, check the chemical components of the sample. The chemical components of the sample must be compatible to the wetted materials of the conductivity cell body. All HORIBA conductivity cells are glass-body, except 9382-10D.
4. Temperature Sensor
Conductivity is highly temperature-dependent. A conductivity cell with built-in temperature sensor allows simultaneous measurement of conductivity and temperature. All HORIBA conductivity cells have built-in temperature sensor, except 3573-10C and 3574-10C.
The electrodes of conductivity cells are coated with a layer of platinum black. The platinum black creates a higher effective surface area for the electrodes and eliminates polarization error. If the platinum black is dry, dirty, or peeled off, it will adversely affect the conductivity measurement.
A clean conductivity cell is necessary in performing an accurate conductivity measurement. The choice of cleaning solution should effectively remove all contaminants based on sample tested without damaging the conductivity cell.
Clean the part of conductivity cell in contact with sample using the appropriate solution and then rinse it thoroughly with clean water. Abrasive objects should never be used in cleaning. A piece of cotton soaked in a cleaning solution can be used with caution.
Conductivity cells should be clean before they are stored for any length of time.
29 June 2017, Rev. 0
Do you have any questions or requests? Use this form to contact our specialists.
HORIBA Advanced Techno, Co., Ltd.
31, Miyanonishi-cho, Kisshoin
Minami-ku Kyoto 601-8306 Japan
Tel: +(81) 75 321 7184
Fax: +(81) 75 321 7291
HORIBA Instruments (Singapore) Pte Ltd.
163 Kallang Way, #08-14,
Mapletree Hi-Tech Park @ Kallang Way,
Singapore 349256
Tel: (65) 6745-8300
Email: laqua(at)horiba.com
HORIBA UK Limited Northampton Office
Kyoto Close,Summerhouse Road,
Moulton Park
Northampton NN3 6FL UK
Tel: +(44) 1604 542 600
Fax: +(44) 1604 542 699
Mail: waterquality(at)horiba.com
HORIBA Instruments Incorporated Head Office
9755 Research Drive
Irvine California 92618 USA
Tel: +1 800 446 7422
Fax: +1 949 468 1790
Mail: labinfo(at)horiba.com
You might also like to know...