

Calcium is one of the essential nutrients taken up by plants from soil for cell wall development. To measure calcium concentration in soil, extraction with 1M ammonium acetate and filtration should be performed prior to placing the filtrate onto the flat sensor of LAQUAtwin calcium ion meter.
All plants need calcium rich soil to grow. The calcium is used by the plant in developing the plant cell walls and membranes. Furthermore, it is a non-leaching mineral (it will stay in the soil) and will improve water penetrability and reduce soil salinity. It is thus helpful to determine the amount of calcium contained in soil. Generally, Atomic Absorption Instruments (AA) or Inductivity Coupled Plasma-Optical Emission Spectrometry Instruments (ICP-OES) are used to measure the amount of calcium ions present in soil. An easier method involves extracting the calcium ion from sample soils with 1 mol/L ammonium acetate (CH3COONH4), and then by using the handy and affordable LAQUAtwin calcium ion meter B-751. The LAQUAtwin Ca2+ meter is used as a quick check to determine the calcium content of soil.
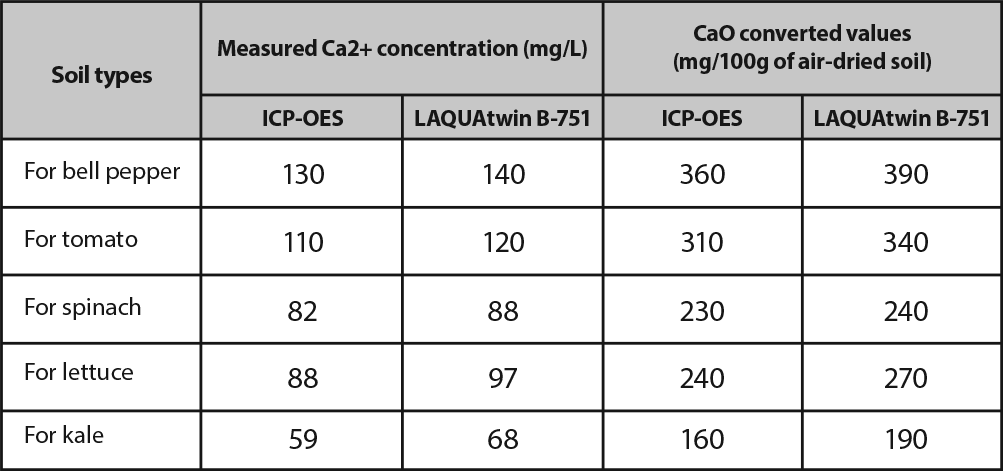
The use of accurate calcium ion testing in controlling the calcium content of soil ensures that the plants which are grown in the soil are given the necessary minerals and can easily absorb water. The table below shows that the results given by the LAQUAtwin Ca2+ pocket meter are comparable to those from Inductivity Coupled Plasma-Optical Emission Spectrometry Instruments (ICP-OES).
The LAQUAtwin Ca2+ pocket meter is small and compact; convenient to carry around in your pocket for quick on-site testing. Its easy-to-use interface is simple for anyone to use the LAQUAtwin Ca2+ pocket meter.
如您有任何疑问,请在此留下详细需求信息,我们将竭诚为您服务。

