

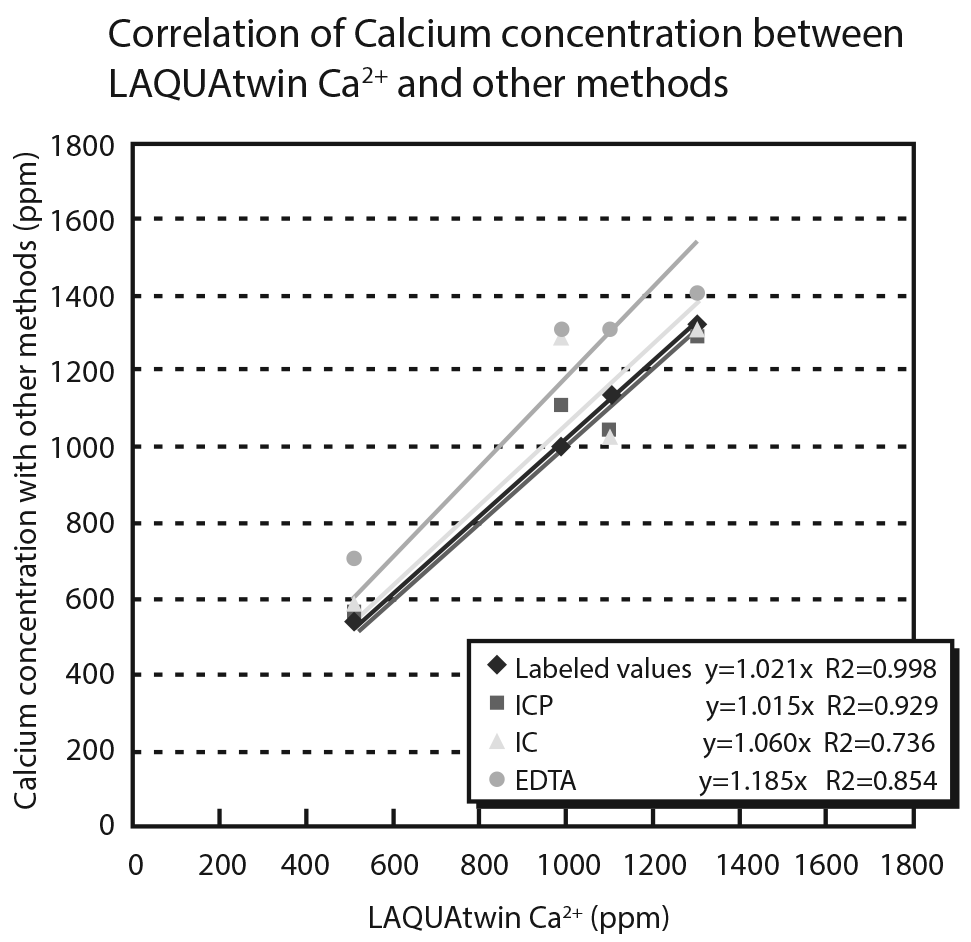
Determining the calcium content of milk and milk beverages helps consumers accurately gauge their calcium intake. Unlike atomic absorption spectroscopy (AAS) and inductively coupled plasma atomic emission spectroscopy (ICP), the LAQUAtwin calcium ion meter offers a simpler method of measuring calcium ion by ionizing protein-bound calcium in sample using acid before analysis.
Measurement of Calcium in Milk and Milk Beverages
It is often necessary to determine the calcium content of milk and milk beverages. This may be done by atomic absorption spectroscopy (AA) or inductively coupled plasma atomic emission spectroscopy (ICP). Alternatively, by ionizing protein-bound calcium using an acidizing pretreatment, the LAQUAtwin Ca2+ can be used to measure the total amount of calcium easily.
The LAQUAtwin Ca2+ meter is used as check to determine the calcium content of milk products before selling to consumers. This is an easy, quick method to check the amount of calcium in lactic products.
Acidify the milk with hydrochloric acid (HCl) to a pH of between 4.3 and 4.6 (which can be determined by use of the LAQUAtwin pocket pH meter). Wait for the solution to precipitate and use the liquid part of the sample for the test. A small sample of the solution is placed on the sensor of the LAQUAtwin Ca2+ and measured. To repeat sampling, wash with diluted soap water and pat dry with a paper tissue.
Acidify the lactic drink with hydrochloric acid (HCl) and use the LAQUAtwin pH meter to confirm it has a pH of about 2. Then, dilute this sample and add tris-hydroxy-aminomethane so that the solution has a pH value between 4.3 and 4.6. Wait for the solution to precipitate and use the liquid to sample. A small sample of the solution is placed on the sensor of the LAQUAtwin Ca2+ and measured. To repeat sampling, wash with diluted soap water and pat dry with a paper tissue.
The use of accurate calcium ion testing in controlling the quality and calcium content of lactic products ensures that consumers are accurately able to gauge their calcium intake. This is especially beneficial for those who are lactose intolerant.
The LAQUAtwin Ca2+ pocket meter is small and compact; convenient to carry around the marketplace for easy on-site testing. Its easy-to-use interface is simple for anyone to use.
1 Internal study by HORIBA labs, 2013
Do you have any questions or requests? Use this form to contact our specialists.
Pocket Water Quality Meters

