

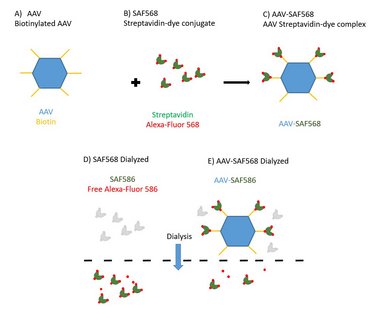
HORIBA is at the forefront of advancing next-generation biotherapeutics and pharmaceuticals, leveraging novel biomolecules and innovative delivery mechanisms to combat diseases. These biotherapeutics, cultivated in cell media, undergo crucial chemical and physical characterization steps for quality assurance during manufacturing and to deepen our understanding during development. From Raman-based protein secondary structure analysis to the rapid infectious titer determination through simultaneous multi-laser Nanoparticle Tracking Analysis, and the A-TEEM fluorescence method for AAV characterization and vaccine batch release, HORIBA provides indispensable insights that matter across the entire biotherapeutic journey, from development to manufacturing.
In the pharmaceutical arena, where small molecule therapeutics offer diverse administration routes, from pills and capsules to inhalation devices, precision is paramount. The quality of these products hinges on knowing component quantities, distribution, and size. HORIBA's comprehensive portfolio of analytical instruments, encompassing cutting-edge Raman microscopy and a full range of particle sizing techniques, accelerates product development, addresses critical troubleshooting questions, and excels in contaminant identification. Additionally, HORIBA's expertise extends to the biotechnology field, where we contribute to all aspects, from R&D to on-line measurement, supporting high-quality, high-efficiency production, and post-use wastewater monitoring in this rapidly evolving industry. These same orthogonal, non‑destructive techniques also underpin advanced pharma forensics—supporting the investigation of unknown impurities, raw material variability, counterfeit products, and formulation inconsistencies—helping manufacturers protect product integrity, ensure regulatory compliance, and reduce risk across the value chain.
A-TEEM Spectroscopy
Rapid Raman Plate Reader – Multiwell Fast Raman screening
QC/QA Raman Laboratory Analyzer
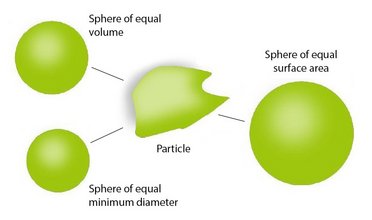
Laser Diffraction and Dynamic Imaging Particle Size and Shape Analyzer
MicroRaman Spectrometer - Confocal Raman Microscope
Simultaneous Multispectral Nanoparticle Tracking Analysis (NTA)
Nanoparticle Analyzer
Fluorescence Lifetime Imaging Platform
Unlocking Innovation Across Disciplines


如您有任何疑問,请在此留下詳細需求或問題,我們將竭誠您服務。