Pharmaceutical and crystallographic samples typically require detailed characterization and analysis to optimize a samples stability, physical properties and indeed general efficacy where an active drug substance is involved.
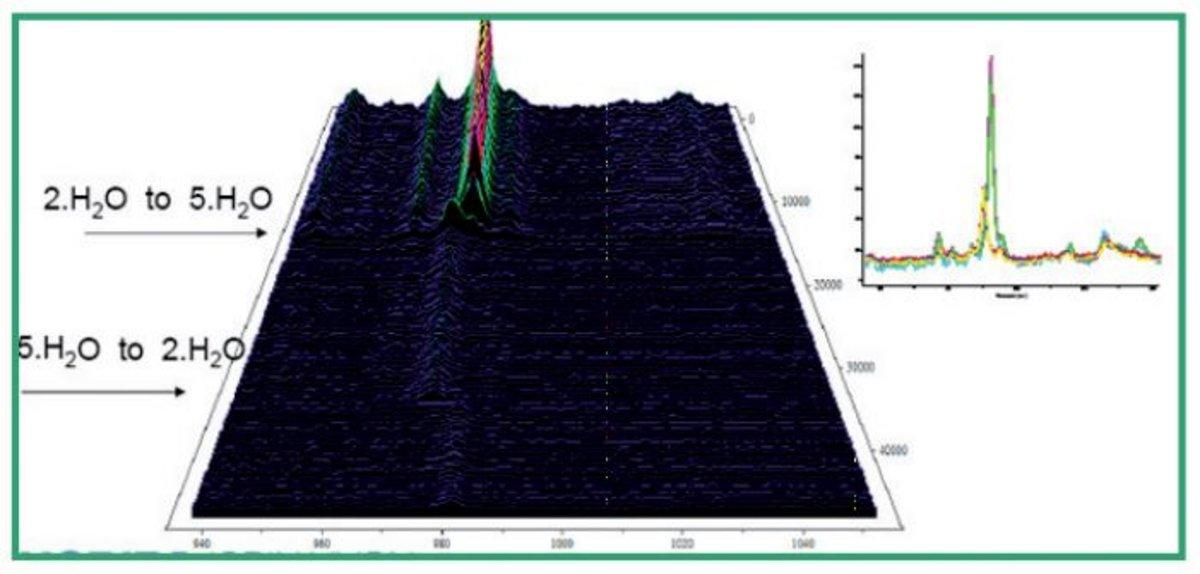
Pharmaceutical and crystallographic samples typically require detailed characterisation and analysis to optimise a samples stability, physical properties and indeed general efficacy where an active drug substance is involved. Furthermore, strict regulatory and manufacturing procedures demand precise characterisation of a solid salt form drug candidate and the possible influence of environmental conditions. The hydration and structure of such samples under varying conditions is important in this full characterisation.
Confocal Raman Microscope
MicroRaman Spectrometer - Confocal Raman Microscope
Confocal Raman & High-Resolution Spectrometer
Sie haben Fragen oder Wünsche? Nutzen Sie dieses Formular, um mit unseren Spezialisten in Kontakt zu treten.