

La distribución del tamaño de partículas de los ingredientes activos y los excipientes es una característica física importante de los materiales utilizados para fabricar productos farmacéuticos. El tamaño, la distribución y la forma de las partículas pueden afectar a las propiedades a granel, el rendimiento del producto, la procesabilidad, la estabilidad y la apariencia del producto final.
La relación entre el tamaño de la partícula y el rendimiento del producto está bien documentada en cuanto a disolución, tasas de absorción y uniformidad del contenido. Reducir el tamaño de las partículas puede ayudar a la formulación de NCE con baja solubilidad en agua. La correcta coincidencia entre el tamaño del ingrediente activo y el tamaño de la partícula excipiente es importante en varios pasos del proceso. El análisis del tamaño de partículas es un componente integral del esfuerzo por formular y fabricar muchas formas farmacéuticas. HORIBA Instruments puede proporcionar tanto las herramientas analíticas como el apoyo técnico necesarios para ayudar a las compañías farmacéuticas a caracterizar sus sistemas de partículas con confianza.
Mira un vídeo de dos minutos detallando cómo HORIBA Scientific se ha asociado con Particle Sciences para ofrecer tecnología y soporte de clase mundial para su negocio CDMO.
Enlace de YouTube: Entrevista con Rob Lee de Particle Sciences
Esta es la técnica de análisis del tamaño de partículas más popular utilizada en la industria farmacéutica. Esta tecnología es rápida, fácil de usar, flexible y repetible. El analizador láser LA-960V2 ofrece ventajas únicas, entre ellas:
Descubre cómo se puede usar el LA-960V2 en tu laboratorio farmacéutico descargando estas notas de aplicación.
Polímeros biodegradables como el ácido poliláctico (PLA) se estudian frecuentemente como posibles portadores para formulaciones de liberación controlada de fármacos activos. Dado que las nanopartículas de PLA suelen estar en el rango de 50 a 500 nm, tanto la difracción láser como la dispersión dinámica de luz (DLS) podrían emplearse para el análisis de tamaño, pero la difracción láser tiene la ventaja de poder detectar agregados superiores a 1 micra. La capacidad única de medir partículas de hasta 30 nanómetros y hasta 5.000 micras hace que el LA-960 esté especialmente cualificado para medir toda la gama de nanopartículas de biopolímeros utilizadas para la administración de fármacos.
Descargar Aplicación Nota 196: Análisis de nanopartículas de biopolímeros usando difracción láser
Un excipiente es una sustancia inactiva utilizada como portadora de los ingredientes activos de un medicamento. Además, los excipientes pueden utilizarse para facilitar el proceso mediante el cual se fabrica un producto. Se utilizaron dos excipientes comunes – estearato de magnesio y celulosa microcristalina – como muestras para las notas de aplicación del desarrollo de métodos húmedos y secos.
Los analizadores de imagen estáticos proporcionan información sobre el tamaño y la distribución de la forma de partículas desde 0,5 hasta 1000 μm. Las principales compañías farmacéuticas utilizan esta técnica para una variedad de aplicaciones, incluyendo la caracterización de activos farmacéuticos, el cribado de excipientos, la validación de métodos de apoyo y la inspección de MDI y DPI.

Detección de partículas grandes mediante un inhalador de polvo seco (DPI) accionado.
Descargar Aplicación Nota 169: Caracterización de partículas de MDIs
Descargar Aplicación Nota 168: Caracterización de partículas de DPI
Ver seminario web: Tamaño de partículas y evaluación Raman de una forma dosificada semisólida
La dispersión dinámica de la luz es la técnica preferida para el análisis del tamaño de partículas submicron. El analizador de nanopartículas SZ-100V2 mide el tamaño de las partículas desde 0,3 nanómetros hasta 8 micras, potencial zeta y peso molecular. El SZ-100V2 puede utilizarse para una variedad de muestras/aplicaciones, incluyendo liposomas, dendrímeros, proteínas, virus y partículas similares a virus.

El analizador de nanopartículas SZ-100V2 mide el tamaño de las partículas, el potencial zeta y el peso molecular.
El estudio de la agregación de proteínas abarca una amplia gama de interacciones y mecanismos. Muchos estudios en esta área investigan la agregación de proteínas mal plegadas, que se cree que son responsables de muchas enfermedades degenerativas. Dado que la agregación suele provocar un cambio físico en el tamaño de la proteína, el análisis del tamaño de partículas ha demostrado ser una técnica experimental útil en este campo.
Descargar Aplicación Nota 221: Análisis preciso de agregación de proteínas
Los liposomas son vesículas de doble capa hechas de fosfolípidos derivados de materiales naturales o sintéticos. Se utilizan principalmente en el campo farmacéutico para el tratamiento de cánceres como portadores de fármacos quimioterapéuticos hacia la zona tumoral. La cantidad de fármaco cargada en los liposomas y su tamaño juegan un papel fundamental en los parámetros farmacocinéticos y farmacodinámicos del fármaco. Por ello, una medición precisa y rápida del tamaño de los liposomas es esencial para sistemas novedosos y efectivos de administración de fármacos.
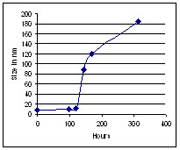
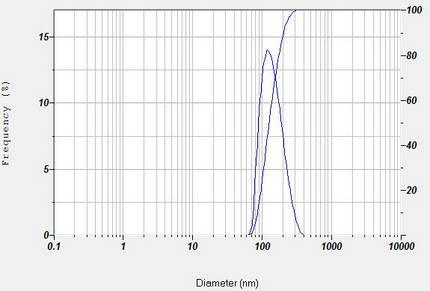
Los datos del SZ-100 que se muestran a continuación muestran la distribución del tamaño de partículas de liposomas disponibles comercialmente.
Distribución del tamaño de partículas para un material liposomal medida por el SZ-100.
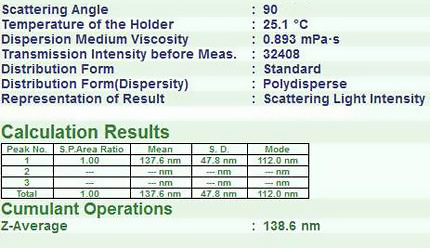
Resultados del tamaño de partícula para el liposoma.
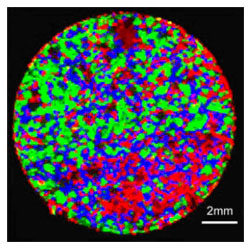
El ARAMIS de la División Raman HORIBA Jobin Yvon proporciona información sobre las propiedades de las partículas, incluyendo tamaño, forma e identificación química dentro de productos finales como tabletas. Estos datos son útiles para investigar la uniformidad del contenido, polimorfismo, espesores de recubrimiento, mapeo de componentes y diferenciación entre fases amorfas y cristalinas.
Ahora, con las opciones de mapeo Raman rápido recién lanzadas, SWIFT™ y DuoScan™, tabletas completas como la tableta de 12,5 mm de diámetro mostrada arriba pueden mapearse en 10 minutos o menos.
Dendrimers
El tamaño de los dendrímeros se mide mediante dispersión dinámica de la luz.
Liposomas
El tamaño de los liposomas se mide mediante dispersión dinámica de la luz.
Virus y partículas similares a virus
El tamaño de los virus y los VLP se mide mediante dispersión dinámica de la luz y difracción láser.
Fabricación y desarrollo de vacunas
Los virus, VLP y exosomas pueden analizarse por tamaño y concentración mediante el Análisis de Seguimiento de Nanopartículas (NTA) multiláser.
Analizador de Distribución de Tamaño de Partículas por Difracción Láser
Análisis Simultáneo de Seguimiento Multiespectral de Nanopartículas (NTA)
Analizador de Nanopartículas
Tiene alguna pregunta o solicitud? Utilice este formulario para ponerse en contacto con nuestros especialistas.